SwissChems — Our Response to The New Yorker and Finnrick Analytics
April 2026
To our valued customers and the research community,
Recent media coverage — including a piece in The New Yorker [1] — has referenced testing results from Finnrick Analytics that characterize several SwissChems products as substandard. We take product quality seriously, and we owe you a transparent account of what these results actually show, who produced them, and why we believe the conclusions drawn are misleading.
We are not asking you to take our word for it. We are presenting the evidence and letting you judge for yourselves.
Too long? Here’s what you need to know.
- Finnrick’s own labs disagree by up to 59% on the same batch. TrustPointe measured our Ipamorelin at 1.53mg. MZ Biolabs measured the same batch at 0.62mg. One lab is wrong. Finnrick published both and averaged them.
- CJC-1295 DAC “54% purity” is analytically incomplete. Our own pre-shipment testing showed this batch at 99%+ purity. MZ Biolabs found three peaks but only identified one of them — classifying 44% of the vial as “unknown” without ever checking what it was. The three-peak pattern is consistent with maleimide hydrolysis, a known degradation process accelerated by improper storage.
- The BPC-157 lab photo shows two compounds on the label. TrustPointe’s own photograph references both BPC-157 and TB-500, but they only tested for BPC-157 and called it underdosed.
- Finnrick is not a lab. It’s apparently a VC-funded startup run from a co-working space by a gaming entrepreneur and a marketing consultant. They charge vendors for premium ratings and hire paid “Gray-Market Whisperers” to infiltrate peptide forums.
- The labs Finnrick uses are not accredited. MZ Biolabs is a 2-person operation. Krause Analytical has a documented 15% measurement bias. Neither holds ISO/IEC 17025 accreditation.
- No chain of custody. Our products sat at Finnrick’s facility for up to two weeks with no documented refrigeration before being shipped to labs. Peptides degrade without proper storage.
- The “lead” and “endotoxin” claims come from a separate, non-public test by Krause Analytical that doesn’t even appear on Finnrick’s own website. We’ve commissioned independent accredited testing to verify.
- SwissChems has operated for 7 years with tens of thousands of repeat customers. We will not pay Finnrick for better ratings. We will prove our quality through independent, accredited testing.
Who Is Finnrick Analytics?
Finnrick Analytics is apparently a venture-capital-backed startup founded in March 2025 — barely one year old [2]. It was founded by Michael Maxwell Carter, a Silicon Valley gaming entrepreneur whose previous ventures include Game Closure (a mobile gaming company) and Hello World (sold to Life360) [3], and Raphaël Mazoyer, a French digital marketing strategist whose prior work involved e-commerce for ASICS and Lacoste [4]. Neither founder has a background in health, pharmaceuticals, or analytical chemistry.
Finnrick is backed by investors including Naval Ravikant (AngelList founder) and Walter Kortschak (early investor in Lyft, Robinhood, and Palantir) [2]. These are financial investors seeking returns — not scientists pursuing public health.

Finnrick’s own About page: Naval Ravikant, Walter Kortschak, Tom Fairfield, Cameron Teitelman — financial investors, not scientists. Source →
Critically, Finnrick operates with structural conflicts of interest that no legitimate testing organization would tolerate:
- Finnrick charges vendors for premium listings, “Launch” programs, and private testing — while simultaneously assigning those same vendors public letter grades. Their Vendor Support page [5] lists paid programs including $279/month premium registration, paid “Launch with Finnrick” testing, and confidential “Private Testing” where results are not published. Their own job listing states the business model plainly: “We fund the mission by supporting vendors who want to win” [6]. Vendors who pay Finnrick are rated by Finnrick. There is no published conflict of interest policy addressing this.
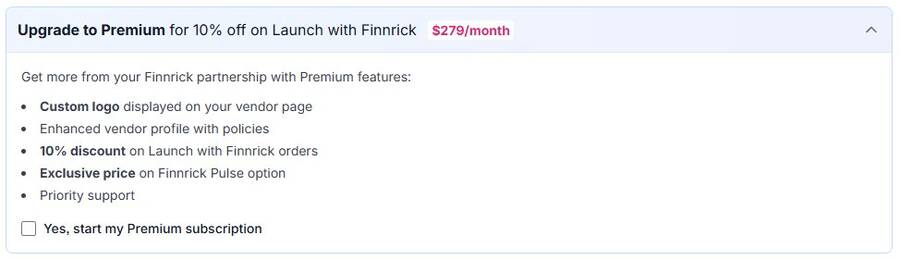
$279/month for “Premium” — includes custom logo on your vendor page, priority support, and discounts on testing. Source →
- Finnrick employs paid community operatives. A public job listing for a “Gray-Market Whisperer” ($85,125–$125,000/year + equity) [6] is remarkably candid about its purpose. The role involves building “a cross-platform content engine that turns HPLC graphs into viral hooks,” creating email sequences that “feel like DMs from a trusted plug, not a newsletter,” maintaining “a real-time intel dashboard tracking every new peptide Telegram drop and Reddit ban wave,” and being “Finnrick’s voice inside every peptide tribe.” The listing describes “debunking a vendor in a 4chan thread” one hour and going “live on TikTok injecting saline for transparency theater” the next — their word, not ours: theater. The listing requires “AI prompt wizardry without sounding like ChatGPT” — meaning AI-generated content deliberately disguised as human. Applicants are told to apply via meme on X with no résumé or cover letter, screening specifically for people who can blend into communities undetected. Under FTC endorsement guidelines, commercial messaging disguised as organic peer communication is deceptive. Finnrick published this job description publicly.

$85k–$125k/year + equity to infiltrate peptide communities. “Transparency theater.” “AI prompt wizardry without sounding like ChatGPT.” View full listing →
- Finnrick holds no ISO/IEC 17025 accreditation, nor do several of the labs it uses. Legitimate testing authorities — ConsumerLab, USP, NSF International — all maintain formal accreditation, published conflict of interest policies, and structural separation between revenue and editorial decisions. Finnrick has none of these. An independent review by Peptide Protocol Wiki documented these concerns in detail [7].
What the Lab Results Actually Show
We have obtained and reviewed the Certificates of Analysis that Finnrick published for our products. The findings raise serious questions — not about our products, but about Finnrick’s testing methodology and the labs they use.
A note on what exists publicly vs. what does not: Finnrick’s public vendor page for SwissChems [19] lists exactly nine tests across three products (2× BPC-157, 4× Ipamorelin, 3× CJC-1295 DAC). The New Yorker‘s claims about lead in BPC-157 and endotoxins in TB-500 come from a separate, non-public submission — the journalist sent samples directly to Krause Analytical (a Finnrick partner lab), and those results were never posted on Finnrick’s public vendor page. We address both the public dataset and the separate Krause claims below.
Ipamorelin 2mg — Four Tests, Two Labs, One Clear Conclusion
Finnrick ordered two vials of Ipamorelin 2mg from SwissChems in the same order (March 19, 2025) and sent both to two different laboratories. This gives us a rare four-way comparison — the same product, same batch, tested by two independent labs:
| TrustPointe Analytics | MZ Biolabs | Discrepancy | |
|---|---|---|---|
| Vial 1 | 1.551 mg (99.57% purity) | 0.87 mg (99.96% purity) | MZ Biolabs reads 44% lower |
| Vial 2 | 1.526 mg (99.97% purity) | 0.62 mg (99.95% purity) | MZ Biolabs reads 59% lower |
Sources: TrustPointe COA SPL-0693 [8]. MZ Biolabs COAs lv7d948 [9] and 5y2qpps [10]. All certificates viewable at finnrick.com/verify.

TrustPointe: Vial 1 = 1.551mg, Vial 2 = 1.526mg (1.6% internal variance)
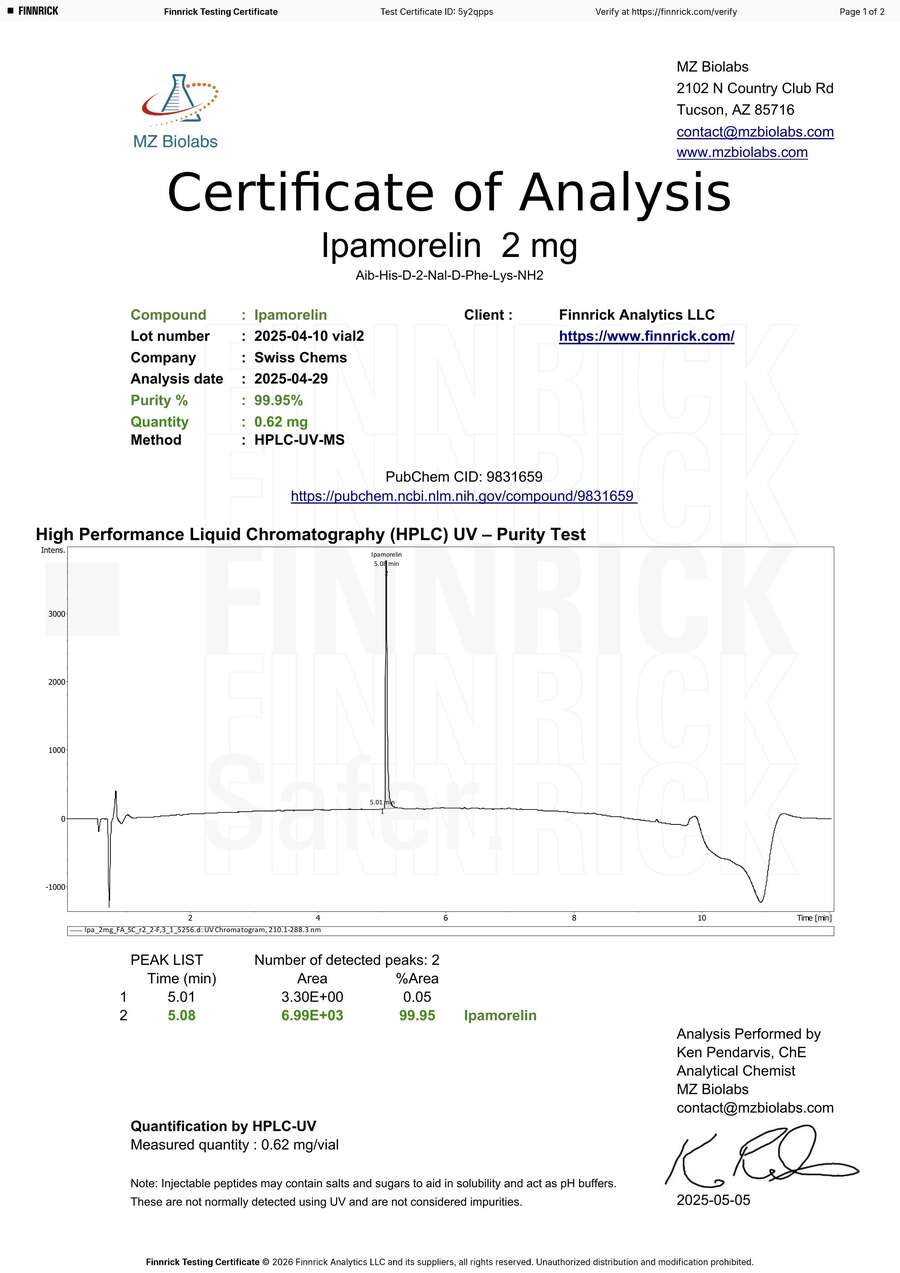
MZ Biolabs: Same batch 2 = 0.62mg — 59% lower than TrustPointe
All four tests confirm the product is ipamorelin. All four tests confirm purity above 99.5%. The disagreement is entirely on quantity — the single metric that drove the poor rating.
The pattern is clear. TrustPointe’s two results agree with each other within 1.6% (1.551 vs 1.526 mg) — the kind of internal consistency you expect from a reliable lab. MZ Biolabs’ two results disagree with each other by 29% (0.87 vs 0.62 mg) and disagree with TrustPointe by 44–59%. When one lab produces internally consistent results and the other does not, the inconsistent lab is the outlier.
Finnrick had this data. They could see that their own labs disagreed by up to 59% on the same batch. Rather than flagging this discrepancy, investigating the outlier, or excluding the anomalous results, they published all four tests and averaged them into our rating. Any credible testing organization would have recognized that a 44–59% inter-lab disagreement on the same sample indicates a laboratory measurement problem, not a product deficiency.
For reference, independent journalist Rory at Gray Market conducted his own investigation — sending four identical vials from the same batch to Finnrick for testing at different labs — and documented a 15% potency discrepancy between Finnrick’s partner labs on the same product [11]. Multiple community members subsequently contacted him reporting further discrepancies with Krause Analytical specifically.
CJC-1295 DAC 2mg — Degradation Artifact or Product Defect? The Lab Didn’t Bother to Check.
MZ Biolabs tested two vials of our CJC-1295 DAC and reported purities of 54.44% and 57.25% [12][13]. Our own pre-shipment testing of this batch showed purity at 99%+. We do not know what happened to these vials after Finnrick received them.
The HPLC chromatograms show three closely eluting peaks at 6.70, 6.78, and 6.85 minutes — separated by fractions of a minute. MZ Biolabs identified only the first peak as CJC-1295 DAC and left the other two uncharacterized, classifying them as impurities by default.
The peak ratios are nearly identical across both vials:
| Vial 1 | Vial 2 | |
|---|---|---|
| Peak 3 (CJC-1295) | 54.44% | 57.25% |
| Peak 4 (unidentified) | 19.31% | 18.60% |
| Peak 5 (unidentified) | 25.02% | 23.18% |
| Peaks 3+4+5 combined | 98.77% | 99.03% |
If peaks 4 and 5 were random impurities, they would not appear in the same ratio across two separate vials. This consistent fingerprint points to a systematic source — either a structural property of the molecule or a uniform degradation process.
CJC-1295 DAC contains a maleimidopropionyl linker on its Lys30 residue — this is the Drug Affinity Complex that gives the molecule its name [14]. It is well established in pharmaceutical bioconjugate chemistry that maleimide groups undergo ring-opening hydrolysis in aqueous conditions, producing closely related species that separate as multiple peaks on HPLC. Published research documents multi-peak chromatographic patterns in the majority of hydrolyzed maleimide-containing compounds. These hydrolysis products are structurally near-identical to the parent molecule — same peptide backbone, slightly modified linker.
Maleimide hydrolysis is accelerated by moisture, temperature, and time. Finnrick held these vials for 19 days with no documented refrigeration before sending them to MZ Biolabs. Fresh CJC-1295 DAC from other vendors consistently tests at 98–99%+ purity with a single dominant peak.
The most likely explanation: our CJC-1295 DAC was 99%+ purity when it shipped — consistent with our own testing. After 19 days of unknown storage conditions at Finnrick’s facility, the maleimide linker partially hydrolyzed into related species. MZ Biolabs then reported those degradation products as impurities — without ever checking whether they shared the same molecular weight as the parent compound.
Both vials confirmed identity via mass spectrometry with a perfect molecular weight match (expected 3645.02 Da, measured 3645.02 Da on both). However, MZ Biolabs ran MS only on the main peak — extracting spectra from the 6.60–6.74 minute window only [12][13]. They never characterized peaks 4 and 5. Any competent analytical chemist encountering three closely spaced peaks on a maleimide-conjugated peptide would run MS on all of them before reporting 54% purity. MZ Biolabs did not.
Reporting “54% purity” without characterizing 44% of the vial contents is not a finding. It is an incomplete analysis. Finnrick published both results as “E” ratings without questioning why a confirmed CJC-1295 DAC product would consistently show exactly 43–46% “impurities” in the same ratio across multiple vials.
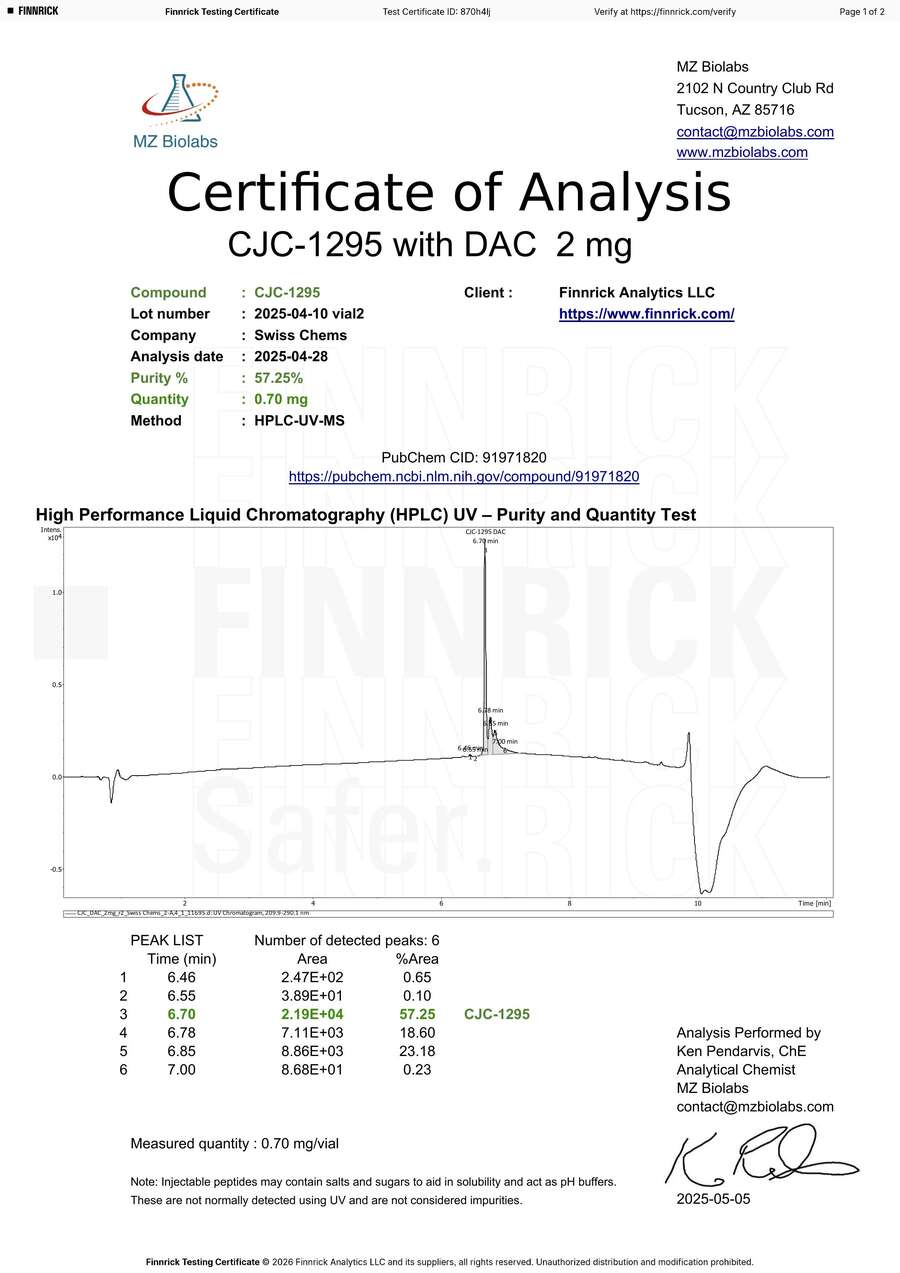
Vial 1: Three peaks at 6.70/6.78/6.85 min — ratio 54:19:25
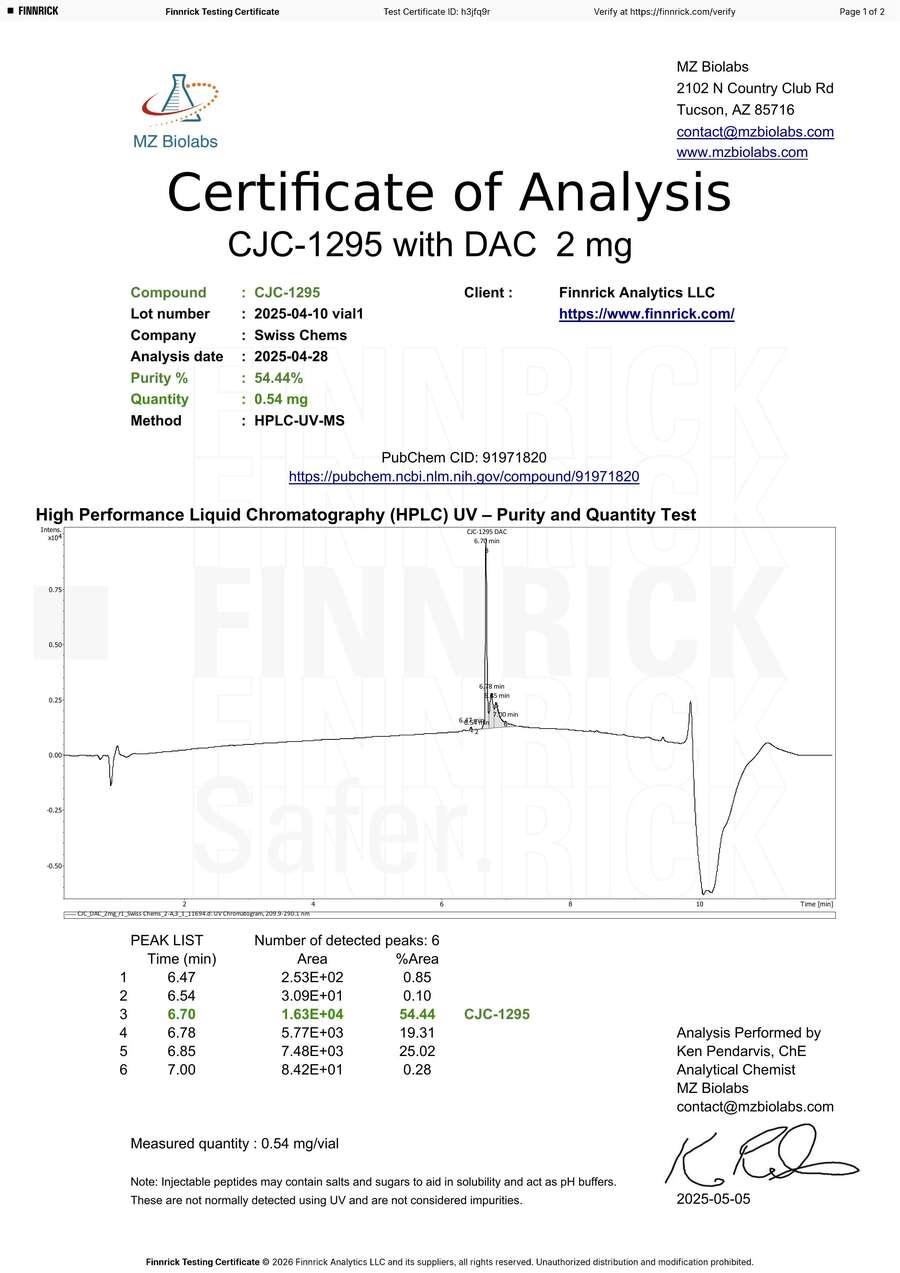
Vial 2: Same three peaks, ratio 57:19:23

Close-up: Three peaks at 6.70, 6.78, and 6.85 min. MZ Biolabs only ran MS on the main peak and never characterized the other two — then reported “54% purity.”
BPC-157 5mg — Tested at the Wrong Specification?
The BPC-157 test was performed by TrustPointe Analytics [15]. The results were reasonable: 99.71% purity, 4.11 mg measured against a 5 mg label claim (82.28% label claim). TrustPointe’s own comment: “Sample is sub potent.”
However, the photograph attached to the Certificate of Analysis — taken by the laboratory and included on page 2 of the COA [15] — shows a sample label referencing both BPC-157 and TB-500. If the tested vial was a combination product, testing only for BPC-157 and reporting the result as a single-compound potency measurement would be an error in test design, not evidence of an underdosed product.
Furthermore, an 82% label claim for a lyophilized peptide is within the range commonly attributable to TFA/acetate counterion salt content, which typically accounts for 15–30% of gross peptide weight [16]. This is an industry-wide labeling convention, not a SwissChems-specific issue.

Red boxes: Lab sample labeled “BPC-157 & TB-500” — but the SwissChems vial only says “BPC-157.”

SwissChems branded vial with original label intact — not relabeled as Finnrick claims.
Lead in BPC-157 — The Krause Analytical Claim
The New Yorker article states that the journalist’s BPC-157 vial “contained lead.” This claim comes from a separate, one-off test submitted directly to Krause Analytical — a Finnrick partner lab — rather than processed through Finnrick’s standard vendor-rating platform. Those specific results do not appear on Finnrick’s public SwissChems vendor page [19] and have not been shared with us in raw form.
This matters for several reasons. Krause Analytical is the same lab that independent investigator Rory at Gray Market documented as producing results 15% lower than every other lab on identical samples [11]. After that investigation was published, multiple community members contacted Rory reporting further discrepancies with Krause specifically. A single unreplicated heavy-metal result from a lab with documented quantification problems — on a sample with no verified chain-of-custody — is not the kind of evidence that should drive a national media narrative.
For full transparency: the public TrustPointe COA for our BPC-157 (May 5, 2025) [15] shows 99.71% purity with no contamination flags in the summary. The attached laboratory photograph references both BPC-157 and TB-500 on the sample label, raising the possibility that the tested sample involved a blended product or a labeling mix-up at the Finnrick or lab level.
Endotoxins in TB-500 — The Missing Product
The New Yorker article also claims that our TB-500 “contained endotoxins.” Finnrick’s public vendor page for SwissChems lists no tests or ratings for TB-500 [19]. The endotoxin claim therefore stems from the same non-public, journalist-direct submission to Krause Analytical. We have no access to that specific COA and cannot verify chain-of-custody, sample handling, or methodology.
Endotoxins — fragments of bacterial cell walls — are a known risk in any non-GMP peptide manufacturing and are a legitimate concern for injectable products. We do not dismiss this. However, a single unreplicated endotoxin result from a lab with documented reliability issues, on a sample with no verified storage or handling protocol, is not sufficient evidence to characterize a vendor’s manufacturing quality.
Chain of Custody — What Happened to Our Products Before Testing?
Finnrick does not operate a laboratory. Their facility is a compact office on the second floor of a co-working space in Austin, Texas. The New Yorker journalist who visited described the scene: a figurine of Goku from “Dragon Ball” on a filing cabinet, scores of vials sitting in trays on a table, and a box stuffed with incoming packages underneath [1]. This is where peptide samples — including ours — are received, handled, and held before being forwarded to partner labs. It is not a climate-controlled analytical facility. Finnrick claims that samples are relabeled before being sent to labs so that “the lab doesn’t know where it’s coming from” [1]. However, the Certificates of Analysis we obtained tell a different story: TrustPointe’s COAs explicitly identify “Swiss Chems” in the client sample ID field, and the attached laboratory photographs show our original SwissChems-branded vials with labels intact [8][15]. The labs knew exactly whose products they were testing.

The TrustPointe lab photograph shows the original SwissChems-branded BPC-157 vial sitting right next to the lab sample container. The vial label — including vendor name and product — was never removed. Finnrick claims samples are blinded before reaching labs. This image, from their own COA, proves otherwise.
There is no published protocol for refrigeration, temperature monitoring, or environmental controls at any point in this process.
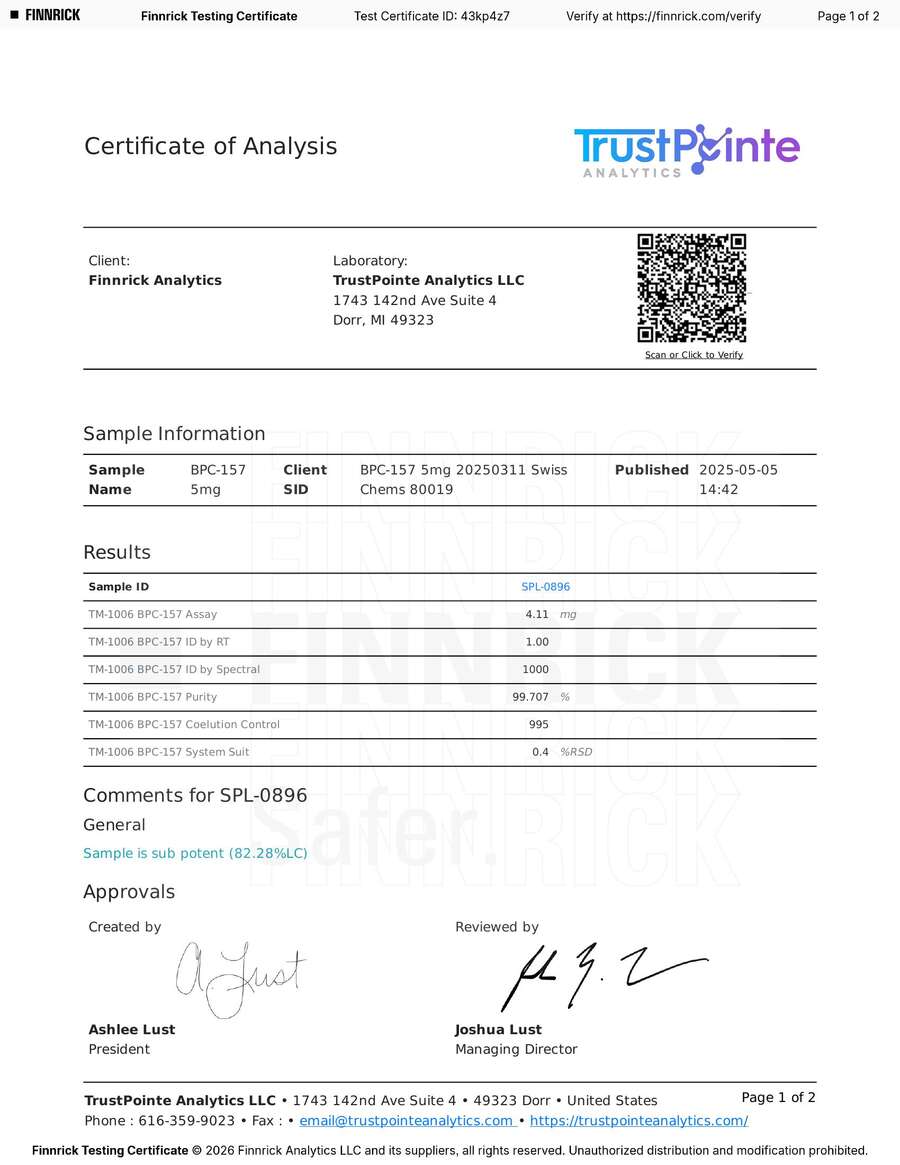
Client SID: “BPC-157 5mg 20250311 Swiss Chems 80019” — the lab knew exactly whose product they were testing.
Peptides are storage-sensitive compounds. Published research shows lyophilized peptides remain stable for years at -20°C, and tolerate controlled room temperature (20–22°C) for weeks to months with an intact seal [17]. But “controlled room temperature” and “a co-working space in Austin, Texas during spring and summer” are not the same thing. At temperatures above 30°C — common in non-climate-controlled spaces, vehicles, and shipping containers — degradation accelerates significantly, particularly when combined with humidity [17][18].
According to Finnrick’s own logistics data, our products sat at their facility for up to two weeks before being sent to a lab. Some tests were not completed until six weeks after purchase. The discrepancy between TrustPointe’s results (tested early April, closer to purchase) and MZ Biolabs’ results (tested late April/early May) is consistent with time-dependent degradation — the later the test, the lower the quantity.
Without verified cold-chain documentation, there is no way to distinguish between a genuinely underdosed product and a properly dosed product that degraded in someone’s filing cabinet.
The Research Chemical Industry — Context Matters
We want to be direct with our customers about something: SwissChems operates in an industry where competitive attacks are a daily reality.
We deal with fake negative reviews posted by competitors on a regular basis. We have been targeted by coordinated campaigns across Reddit, Trustpilot, and other platforms — often by accounts created specifically for that purpose. We have experienced hacking attempts, DDoS attacks on our website, and deliberate efforts to disrupt our operations. This is not unique to us — it is the nature of the research chemical market, where margins are high, regulation is thin, and there is no industry body policing competitive conduct.
We are not suggesting that Finnrick Analytics was hired by a competitor to target us specifically. But we are not alone in questioning their model. Derek Pruski, a prominent peptide community analyst, independently theorized what he called a “shakedown model” in his Substack investigation of Finnrick [23]: publish poor results for established vendors, wait for those vendors to come to the platform to “verify” or get “certified,” and convert them into paying customers. The Peptide Protocol Wiki arrived at similar conclusions in their comprehensive review [7], as did investigator Rory at Gray Market [11], who documented measurable lab discrepancies with his own controlled experiment.
To be clear: SwissChems will not pay Finnrick Analytics a single dollar. We will not purchase a premium listing. We will not enroll in their “Launch” or “Grow” programs. We will not pay for private testing to improve our scores. If their business model depends on vendors paying for better ratings, they will not find a customer in us. Every dollar we would spend on Finnrick will instead go toward independent, accredited testing through publicly recognized laboratories — and we will publish those results for our customers to verify for themselves.
A testing platform that accepts anonymous sample submissions, employs paid forum operatives, and publishes results without verified chain-of-custody documentation is a tool that can be weaponized — whether or not that is its stated intent.
Our customers should evaluate all third-party claims about our products — positive or negative — with this context in mind.
Our customers should also understand how this industry actually works. Anyone can open a peptide company tomorrow. There are no barriers to entry, no licensing requirements, no mandatory testing. A new vendor can launch a website, spend a few thousand dollars on affiliate commissions and paid reviews across Reddit, YouTube, and TikTok, and within weeks appear to be a “top-rated brand” with glowing testimonials and impressive-looking COAs. Credibility in this market is not earned — it is purchased. Finnrick is simply a newer version of the same dynamic: instead of buying reviews, vendors buy premium listings and “Launch” programs from the platform that rates them. The packaging is different. The mechanism is the same. SwissChems has been in business for seven years not because we bought our reputation, but because our products brought customers back. That is the only form of credibility that cannot be faked.
What We Are Doing
- Independent verification. Every batch we sell is tested before it ships. We currently work with private analytical laboratories for our internal QC. SwissChems is actively transitioning from private lab testing to publicly recognized, renowned testing laboratories. This transition is already underway and will be completed in the very near future — so that our customers can verify results for themselves rather than relying on our word alone.
- Batch identification. Since January 2026, we have been rolling out visible batch identifiers across all vial labels, SKU by SKU. This rollout will soon be completed across our entire product line, enabling full traceability and independent verification for every product we ship.
- Transparency. We will publish all results — favorable and unfavorable — on our website as they become available. We invite customers to submit our products for independent testing and share the results publicly.
Our Commitment
SwissChems has been operating continuously since 2019 — seven years serving the research community. In that time, we have fulfilled tens of thousands of orders to researchers and institutions worldwide. Our customer base has grown consistently year after year, driven overwhelmingly by repeat purchasers.
We state this not as a marketing claim but as a data point. A vendor selling ineffective products does not retain customers for seven years. Researchers who obtain poor results from a compound do not reorder it. The research community is sophisticated, critical, and unforgiving of vendors who fail to deliver. Our continued growth is the most robust form of quality evidence available — real-world outcomes from thousands of independent users over the better part of a decade, across dozens of product lines.
This track record does not make us immune to criticism, nor does it exempt any individual batch from scrutiny. But it provides essential context when evaluating claims from a one-year-old startup that tested 9 samples using unaccredited labs with documented reliability problems.
We will not allow our reputation to be defined by an apparently VC-funded startup with no accreditation, documented conflicts of interest, paid astroturfers, and labs that disagree with each other by 59% on the same sample.
We encourage our customers to ask questions, request COAs, and hold us accountable. That is how trust is built — not through opaque rating systems designed to monetize fear.
Thank you for your continued confidence.
SwissChems
swisschems.is


